NEWSLETTER: May 2026
Skylims QC insights Part 4 - What value do I get out of my QC setup in Skylims?
Edition 6
5-minute read
Over the past three issues we covered real lab problems like how to see your QC status, how to do your QC configuration and how QC connects to patient results. In this final edition of QC insights, we will show you real data on the value a configured QC plan delivers, how Skylims supports you during audits for QC and lastly, we tie it all together by ending where we started - with the QC Dashboard.
The Impact: What the Data Shows
QC configured in Skylims does more than keep your accreditation in good standing. It creates measurable improvements in efficiency, turnaround time and result reliability. It can also contribute to accuracy of financial reports, e.g. calculating cost per billable / reagent volumes vs. test volumes, giving you valuable insights into your procurement statistics.
Here is what Skylims QC data from labs in active use shows:
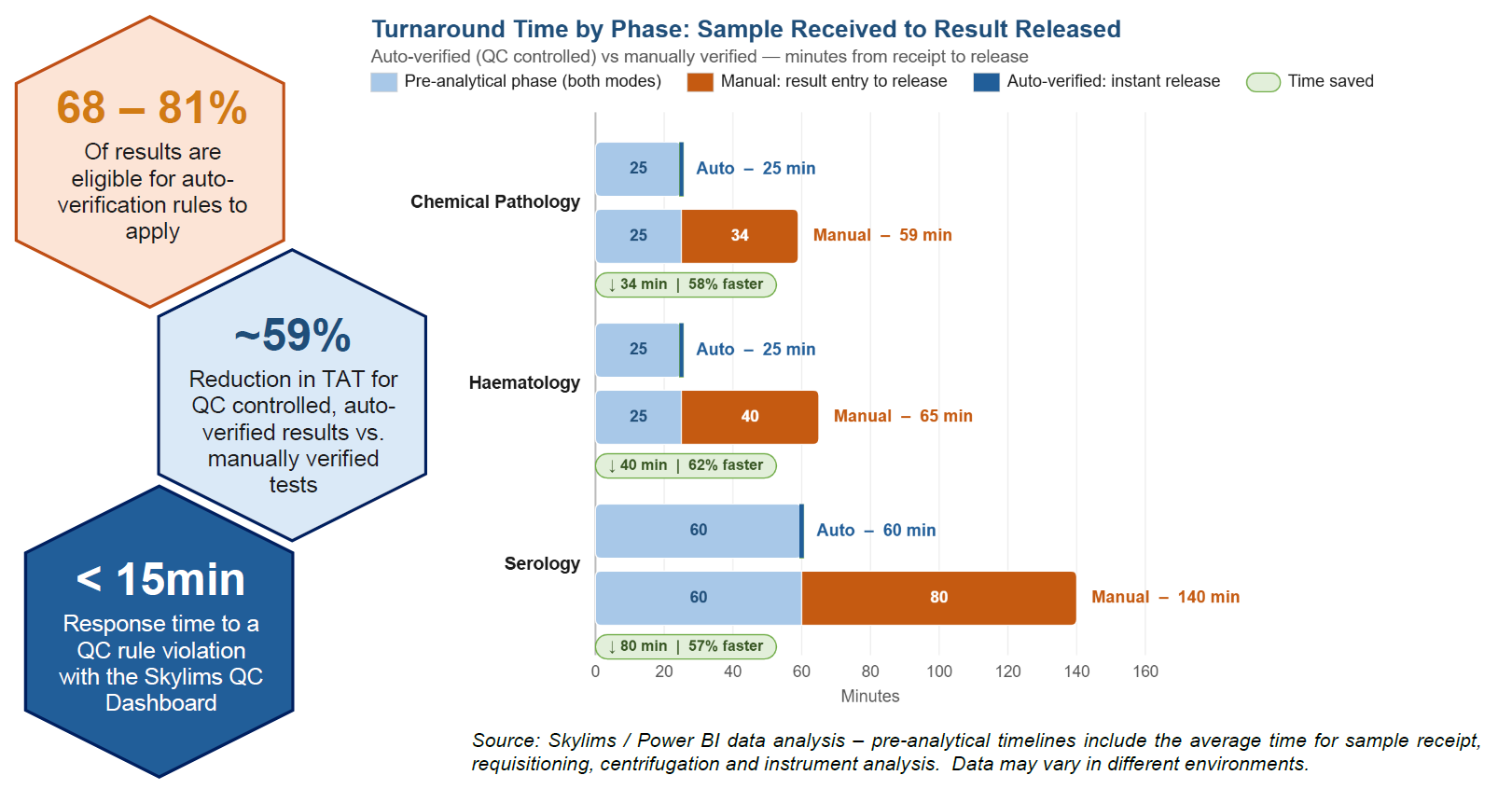
QC configuration is a powerful tool that can be leveraged to make Skylims do the heavy-lifting for you - in a busy lab environment where multi-tasking is a given, automation is a shining beacon to reduce the workload. Automated workflows shift your team's focus from routine tasks to the exception cases that truly require their input and expertise. When QC is added to automation, risk factors go down and result reliability goes up. Focus moves towards what matters most – giving special attention and care to patient results.
💡 Quick Tip:
The Analyte State feature introduced in the previous issue is the gateway to auto-verification. When an analyte is under control, Skylims can automatically release results without manual intervention, cutting turnaround time and freeing your team to focus on exceptions rather than routine sign-offs.
How does Skylims QC help me gather evidence for my next audit?
SANAS assessors use form F 243-02 to assess ISO 15189 compliance during site visits. For QC, assessment covers document review, records review and witnessing. The table below maps every QC-relevant requirement in section 7.3.7.2 to the Skylims feature that produces the evidence.
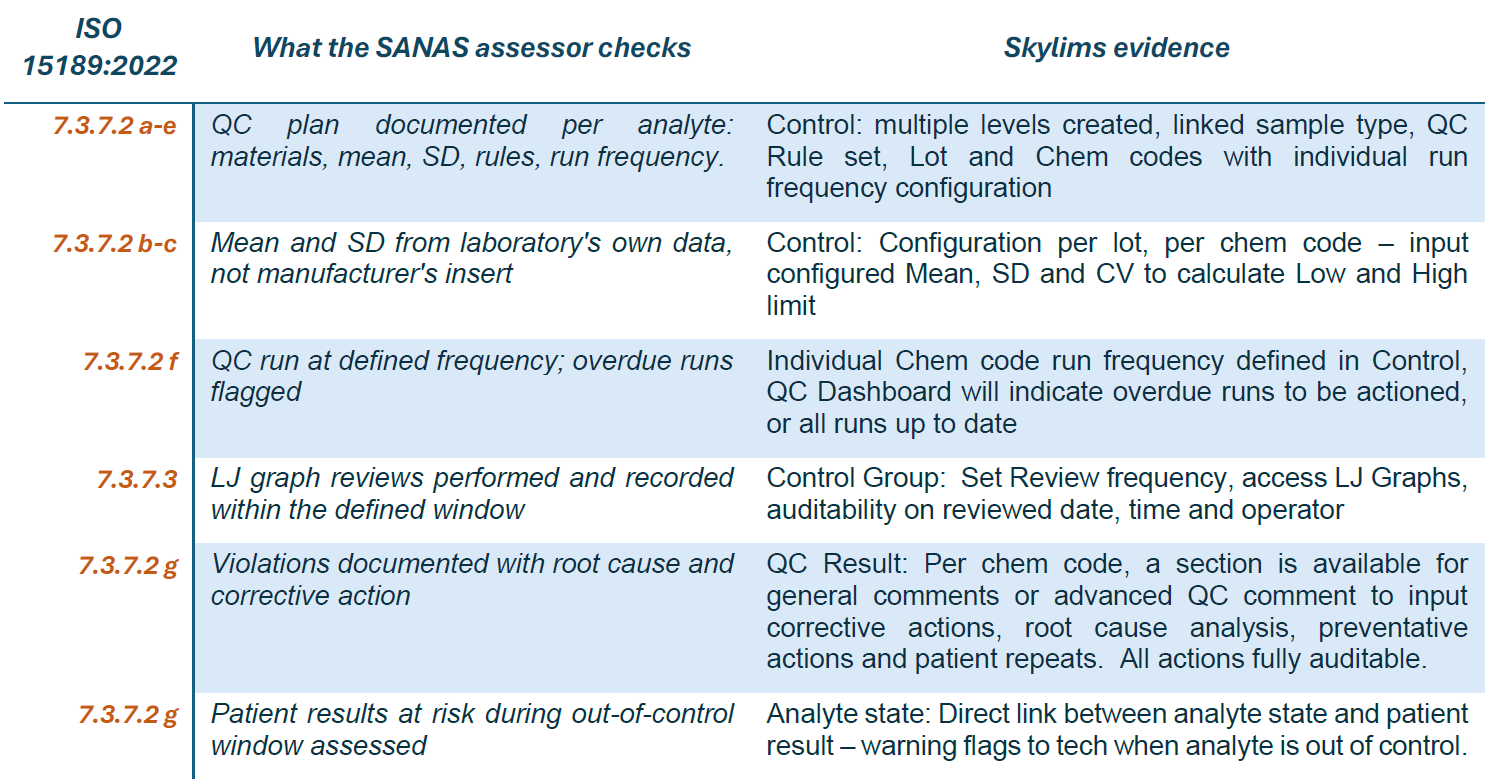
* Source: ISO 15189:2022 | SANAS F 243-02 (2023). *
The full picture: Back to the QC Dashboard
In Issue 1 and Issue 2, the QC Dashboard was the answer to one question: What is the status of my QC? Now you know what sits behind each category. The configuration that drives it. The patient outcomes that depend on it and the possibilities that lie behind it. The dashboard has not changed, but what it means to you, has.

We hope you enjoyed the QC insights series!
| Coming up Next: | Need help with your QC configuration? |
|---|---|
| We answer the question 'How do I make sure nothing slips through?' by exploring the Skylims Task functionality and how responsibility assignment, improved task tracking and greater visibility can transform daily operations. No more sticky notes or forgotten follow-ups; Skylims Tasks bring accountability and clarity into your process. |
Contact the Mukon team for expert guidance on your QC setups. Email: support@mukon.co.za Tel: 012 450 0240 |
Warm regards,
The Mukon Team
References
- ISO 15189:2022. Medical Laboratories: Requirements for Quality and Competence. Clauses 6.6.3, 7.3.7.2, 7.3.7.3. https://www.iso.org/standard/76677.html
- SANAS. F 243-02: Technical Requirements for Medical Laboratories and Blood Transfusion Facilities (ISO 15189:2022). 2023. https://www.sanas.co.za/Publications%20and%20Manuals%20Files/F%20243-02.pdf
- CLSI. Statistical Quality Control for Quantitative Measurement Procedures. 4th ed. CLSI guideline C24. CLSI; 2016. https://clsi.org/media/1365/c24ed4_sample.pdf
- Gruber L, Hausch A, Mueller T. Internal Quality Controls in the Medical Laboratory. Diagnostics. 2024;14(19):2223. https://pmc.ncbi.nlm.nih.gov/articles/PMC11475633/
- Westgard JO et al. A multi-rule Shewhart chart for quality control in clinical chemistry. Clin Chem. 1981;27(3):493-501.
- ASCLS. 'Do No Harm: Diagnostic Errors and the Laboratory.' https://ascls.org/do-no-harm-diagnostic-errors-laboratory/ 7. Plebani M. Ann Clin Biochem. 2010. DOI: 10.1258/acb.2009.009222
- Wieringa S et al. J Patient Safety. 2023. PMC10662575. https://pmc.ncbi.nlm.nih.gov/articles/PMC10662575/